|
3/24/2024 0 Comments Neils bohr atom model However, the idea of quantized angular momentum has been retained. Modern atomic theory does not allow subatomic particles to be treated in the same way as large objects, and Bohr's reasoning is somewhat discredited. The wavelength of a particle is given by h/ mv, so nh/ mv = 2π r, which leads to mvr = nh/2π. 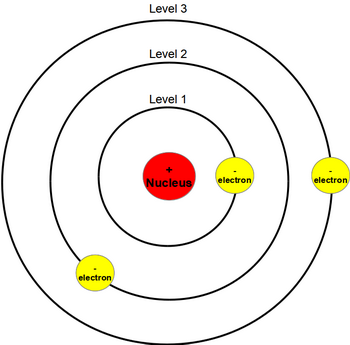
Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. nλ = 2π r, where λ is the wavelength and n a whole number. In 1913, a Danish physicist, Niels Bohr (18851962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Each orbit has to have a whole number of wavelengths around it i.e. The idea of quantized values of angular momentum was later explained by the wave nature of the electron. This theory gave good results in predicting the lines observed in the spectrum of hydrogen and simple ions such as He +, Li 2+, etc. Each permitted value of n is associated with an orbit of different radius and Bohr assumed that when the atom emitted or absorbed radiation of frequency ν, the electron jumped from one orbit to another the energy emitted or absorbed by each jump is equal to hν. can only have certain values, each of which is a multiple of n. This means that the angular momentum is quantized, i.e. The modern model of the atom is based on quantum mechanics. The model of Niels Bohr (18851962) for the atom is since long just the one and only conception for atoms of the vast majority of educated people. 
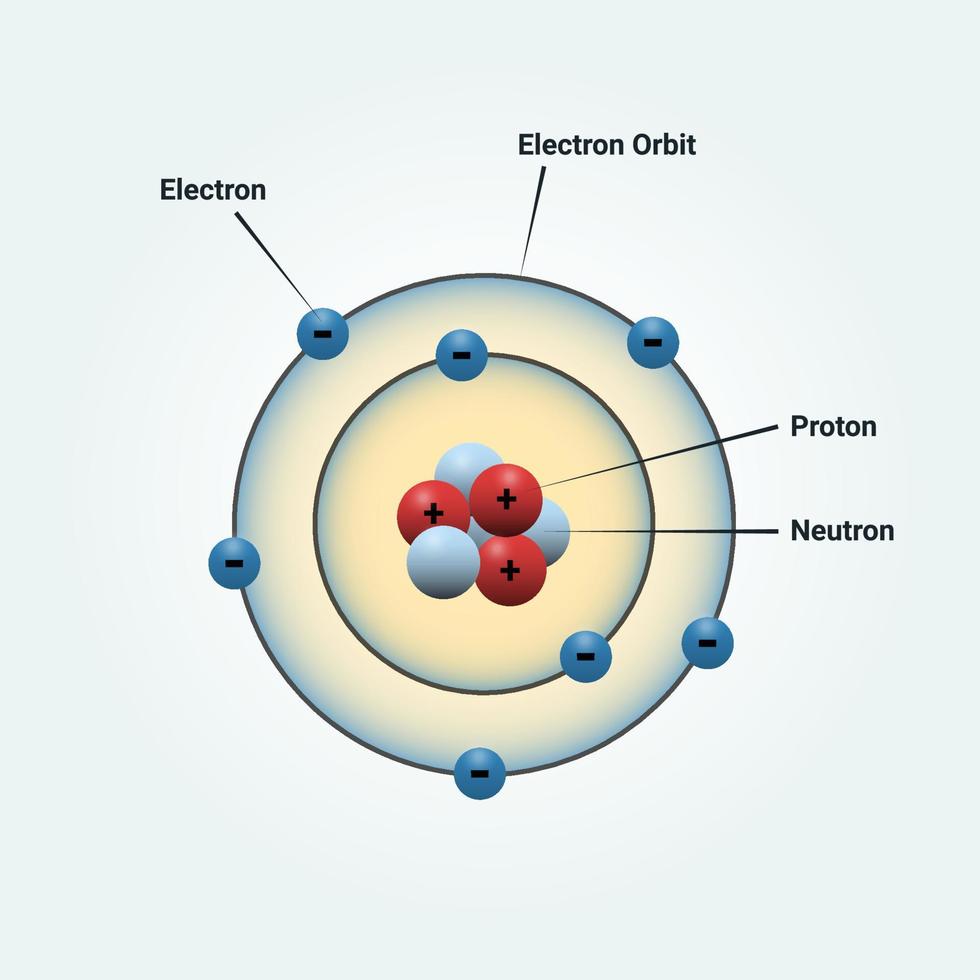
Because the Bohr Model is a modification of the earlier Rutherford Model, some people call Bohr's Model the Rutherford-Bohr Model. Bohr proposed that electrons could only occupy orbits in which this angular momentum had certain fixed values, h/2π, 2 h/2π, 3 h/2π,… nh/2π, where h is the Planck constant. Niels Bohr proposed the Bohr Model of the Atom in 1915. The angular momentum of the electron would then be mvr. He assumed that a single electron of mass m travelled in a circular orbit of radius r, at a velocity v, around a positively charged nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
